Parkinson’s Disease
Can I Get Social Security Disability Benefits for Parkinson’s Disease?
- About Parkinson’s Disease and Disability
- How to Get Disability Benefits for Parkinson’s Disease by Meeting a Listing
- Residual Functional Capacity Assessment for Parkinson’s Disease
- Getting Your Doctor’s Medical Opinion About What You Can Still Do
If you have Parkinson’s disease, Social Security disability benefits may be available. To determine whether you are disabled by your Parkinson’s disease, the Social Security Administration first considers whether it is severe enough to meet or equal a listing at Step 3 of the Sequential Evaluation Process. See How to Get Disability Benefits for Parkinson’s Disease by Meeting a Listing. If you meet or equal a listing because of your Parkinson’s disease, you are considered disabled. If your Parkinson’s disease is not severe enough to equal or meet a listing, the Social Security Administration must assess your residual functional capacity (RFC) (the work you can still do, despite the Parkinson’s disease), to determine whether you qualify for disability benefits at Step 4 and Step 5 of the Sequential Evaluation Process.
About Parkinson’s Disease and Disability
Parkinson’s disease is a common, chronic and progressive neurological disorder caused by deficiency of the neurotransmitter dopamine in an area of the brain called the basal ganglia. The peak age of onset is in the mid-fifties, but the possible range of onset in adults is much wider and the juvenile form can start in childhood or adolescence. Both men and women are affected. Primary Parkinson’s disease and most other forms of parkinsonism are incurable. Attempts at brain cell transplantation, human and animal, have been less than satisfactory. The major current treatment involves replacement of dopamine, as well as drugs to treat associated neurological problems that may be present. The major signs and symptoms of Parkinson’s disease are resting tremor, muscle rigidity, bradykinesia (slowed movement) and hypokinesia (decreased movement), autonomic nervous system dysfunction, and postural instability and abnormal gait. See Residual Functional Capacity Assessment for Parkinson’s Disease.
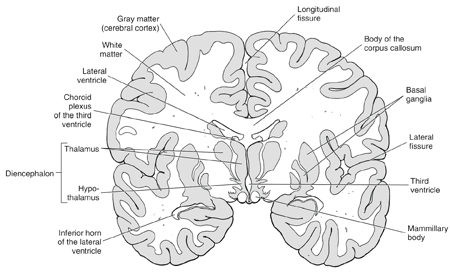
Coronal View of the Brain
Parkinson’s disease is caused by deficiency of the neurotransmitter dopamine in an area of the brain called the basal ganglia. About 80–90% of the dopamine has to be depleted before symptoms appear. The basal ganglia consist of groups of distinctly identifiable cells working together deep under the cerebral hemispheres; such groups of cells are called nuclei. The dopamine deficit is particularly significant in a special group of basal ganglia cells (nucleus) known as the substantia nigra. The dopamine produced in the substantia nigra activates nerve tract projections into neighboring basal ganglia cells, which need input from the substantia nigra to function properly in their important functions related to movement control. Thus, Parkinson’s disease is principally, although not exclusively, a movement disorder.
Neurotransmitters like dopamine are chemicals which jump the synaptic gaps between brain cells, and are the way the brain communicates with itself. The number of neurotransmitter chemicals used by the brain is unknown, but probably exceeds several hundred. In many instances, their function is poorly understood, but dopamine is an important neurotransmitter that has been extensively studied and is clearly very important in many other aspects of brain function besides its role in the basal ganglia.
In the figure below, a normal positron emission tomography (PET) scan shows normal basal ganglia uptake of the D2 Dopamine Receptor Agonist C11 Raclopride, after 30–50 minutes of continuous infusion. Neuroimaging can now provide precise information about deep brain structures that was impossible to obtain in the past.
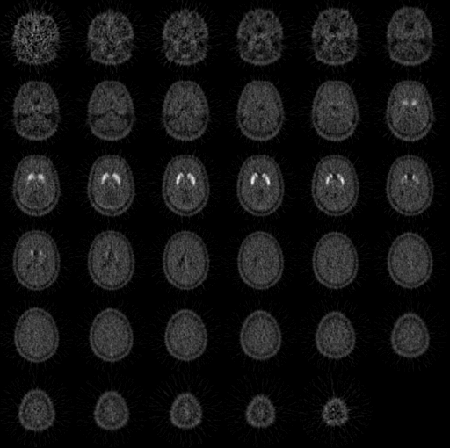
PET Scan Showing Functional Dopamine Cells in Basal Ganglia
Forms of Parkinson’s Disease
Parkinson’s disease is usually taken to mean the disorder is of unknown cause (idiopathic), and is the most common cause of parkinsonism. It is also called primary Parkinson’s disease and primary parkinsonism. Parkinsonism abnormalities associated with other diseases are known as secondary Parkinson’s disease or parkinsonian disorders. To make matters more confusing, Parkinson’s disease is sometimes meant to refer to the idiopathic form, and parkinsonism is used to refer to secondary Parkinson’s disease. And worse than that, some neurologists have various other classification schemes.
Types of parkinsonism include:
- Primary parkinsonism (primary Parkinson’s disease, juvenile parkinsonism).
- Hereditary parkinsonism –Hereditary neurological disorders which can produce parkinsonism because of damage to basal ganglia structures (Huntington’s disease, Wilson’s disease, olivopontocerebellar atrophy (OPCA)).
- Parkinsonism-plus syndromes –Disorders in which parkinsonism is one of the features of some other neurodegenerative diseases. In these disorders, parkinsonian signs and symptoms do not respond as well to treatment and they have a worse prognosis than Parkinson’s disease. (Shy-Drager syndrome (SDS), Alzheimer’s disease, Pick’s disease).
- Secondary parkinsonism –Disorders in which parkinsonism is acquired, rather than being the result of some intrinsic neurological disorder. Some examples are given below. (Drug-induced types are usually reversible; toxic damage is usually irreversible.)
- Drugs (some antipsychotic medications, because they block dopamine receptors (phenothiazines, haloperidol); lithium, reserpine, alpha-methyldopa; antiemetic (anti-vomiting) drugs, such as metoclopramide and prochlorperazine).
- Toxic substances (carbon monoxide; MPTP (a form of synthetic street heroin that destroys basal ganglia cells); alcohol (ethanol and methanol); mercury; cyanide).
- Brain trauma, such as from boxing (encephalopathia pugilistica).
- Vascular disease (multi-infarct dementia).
- Infectious (AIDS encephalopathy, transmission spongiform encephalopathies [prion diseases] like Creutzfeldt-Jakob disease, subacute sclerosing panencephalitis, post-encephalitic parkinsonism).
- Brain tumors.
Signs and Symptoms of Parkinson’s Disease
The major signs and symptoms of Parkinson’s disease are resting tremor, muscle rigidity, bradykinesia (slowed movement) and hypokinesia (decreased movement), autonomic nervous system dysfunction, and postural instability and abnormal gait. Other possible signs and symptoms include decline in cognitive function (e.g., memory loss), emotional disturbance (e.g., depression and anxiety), akathisia (involuntary urge to move), and sleep disturbance (caused by muscle cramps and abnormal body positioning known as dystonia).
Resting Tremor
A oscillatory tremor may affect the arms, legs, face, or tongue. A different disorder often associated with aging—an essential tremor (ET)—causes a head tremor, which would be an unusual finding in Parkinson’s disease. However, the presence or absence of a head tremor is not a reliable way of discriminating the two disorders, because both may easily occur in the same person. Differentiation of the diagnosis is assisted by the fact that—unlike Parkinson’s disease—ET may produce a tremulous voice, rarely involves the lower extremities, and can be improved with the use of alcohol and some drugs, e.g., propranolol (Inderal). Also, essential tremor tends to worsen with activity.
The tremor of Parkinson’s disease may begin on one side of the body, but will progress to the other side as the disease advances. The most notable tremor is often in the hands, where the oscillations produce a repetitive motion that has been classically described as “pill-rolling,” because it is the kind of movement that would be present if a person were rolling something between the index finger and thumb. Forearm tremors are more of a rolling motion oscillation, first one way and then the other (pronation-supination motion). Stress, fatigue, and excitement may cause the tremor to worsen. The resting tremor of Parkinson’s disease is important, because it is an easily identifiable movement characteristic of the disorder. However, Parkinson’s disease can also produce a postural tremor either during use of the hands or evident when the arms are extended out from the body. The tremor of Parkinson’s disease results from the lack of sufficient dopamine to oppose the actions of another important neurotransmitter known as acetylcholine.
Muscle Rigidity
Muscle rigidity is characterized by stiffness and difficulty bending the extremities. In advanced stages of Parkinson’s disease, difficulty in bending the limbs—such as at the wrist—was classically described as like trying to bend a lead pipe. Hence, the term lead pipe rigidity is used to describe this abnormality. Such rigidity is severe enough that examining doctors can detect it on physical examination.
If a tremor is also present, then the individual is likely to have what is called cog wheel rigidity, because when the examining doctor moves the wrist or elbow joint, it moves and stops repetitively, like a cog wheel (ratchet wheel).
Rigidity results in the patient feeling like their limbs are weak and clumsy. Functional loss from rigidity can be marked.
Bradykinesia and Hypokinesia
Bradykinesia means slowed movement, and hypokinesia means decreased movement. Both are a part of the parkinsonian syndrome. In bradykinesia, there is slowness in starting a task, whether getting out of a chair or taking a first step in walking. In the matter of walking, the individual characteristically has the most difficulty starting their gait. In bradykinesia, all activities of daily living (dressing, bathing, eating, etc.) are affected by increased slowness in their performance. The hypokinesia manifests as minimal movement in activities. Bradykinesia can easily be demonstrated on physical examination by obviously slow finger-tapping speed, along with the decreased range of movement characteristic of hypokinesia.
Bradykinesia/hypokinesia also affects the facial muscles, resulting in the impoverished facial expressions known as masked facies (hypomimia). There is an associated decrease in blinking frequency, so that the individual may give the impression of staring. In advanced disease, even swallowing can be difficult (dysphagia). Eating is slowed, and drooling (sialorrhea) may occur. The handwriting is very small and painstakingly executed. The voice is affected: softer speech, poorly articulated speech (dysarthria), a poor range of speech modulation (monotone speech) and hoarse speech may be present.
Advanced parkinsonism results in a phenomenon known as freezing, which is characterized by transient inability to start or change some type of movement. Freezing is a further extension of the severity of bradykinesia/hypokinesia. For example, the individual may suddenly stop walking—freeze in place—when they enter a narrow passage or attempt to turn. But freezing can occur with other types of movements, such as with use of the hands. It takes a few moments to get the movement started again.
Autonomic Nervous System Dysfunction
The autonomic nervous system (ANS) is that part of the nervous system which functions partly or wholly outside of conscious awareness or control. Important examples include regulation of blood pressure, pupillary size, salivation, bladder and bowel functions, erectile function, sweating, piloerection (hair standing up), pallor or flushing, and heart rate. The ANS is divided into the sympathetic and parasympathetic nervous systems. It has been demonstrated that Parkinson’s disease can cause a decrease in available nerve endings in the sympathetic nervous system, particularly the heart. Showing a decrease in sympathetic nerve endings in the heart and other organs, using fluorodopamine with positron emission tomography (PET), implies a sympathetic dysfunction also exists in the rest of the arterial system.
Probably the most severe consequence of sympathetic nervous system dysfunction is orthostatic hypotension. Orthostatic hypotension refers to a drop in blood pressure on changing from a sitting or lying-down position to an upright position. Normally, the sympathetic ANS causes muscles in the walls of arteries to constrict upon standing so that blood pressure can be maintained under the added force of gravity, and the heart must also adapt to such vascular changes. Orthostatic hypotension can cause dizziness and even loss of consciousness with falling. In fact, damage to the sympathetic ANS is present even when orthostatic hypotension is not manifested. This explains numerous other symptoms that may be present in parkinsonism, including gastrointestinal symptoms (especially constipation), abnormal sensations (cold, burning, pain, numbness), oily skin and sweating (sweating can be severe), sexual dysfunction (e.g., impotence), and bladder problems (increased urge, difficulty starting).
Postural Instability and Abnormal Gait
The previously described abnormalities conspire to produce a flexed (stooped) posture and a slow, shuffling gait. Starting to walk is the most difficult, characteristically starting off with small steps. Freezing may occur while walking and falls are frequent, as a result of poor postural reflexes. A festinating gait may be encountered in which small, faster and faster steps are taken in an attempt to prevent falling forward. Festination is an involuntary tendency to fall forward (propulsion) or backward (retropulsion).
Treatments for Parkinson’s Disease
Drug Treatments
Drugs used to treat parkinsonism can be generally classified as:
- Drugs which replace dopamine:
- Levodopa (L-Dopa) – Enters the brain and then is converted to dopamine by the enzyme dopa-decarboxylase.
- Carbidopa/Levodopa (Sinemet) – Carbidopa inhibits dopa-decarboxylase to decrease side-effects that occur when some of the levodopa is converted to dopamine before reaching the brain. These side-effects are nausea, hypotension, confusion, hallucinations, and dyskinesia. Benserazide is another drug that acts like carbidopa. Benserazide/Levodopa is marked under the name Madopar.
- Drugs which mimic the action of dopamine (activate dopamine receptors although not themselves dopamine):
- Bromocriptine (Parlodel)
- Pergolide (Permax)
- Ropinirole (Requip)
- Pramipexole (Mirapex)
- Cabergoline (Dostinex)
- Indirect agonists (drugs that indirectly stimulate dopamine receptors):
- Amantadine (Symmetrel)
- Monoamine oxidase B inhibitor:
- Selegiline (Deprenyl, Eldepryl)
- Drugs which inhibit the action of acetylcholine:
- Trihexiphenidyl (Artane)
- Biperiden (Akineton)
- Benztropine (Cogentin)
- Orphenadrine (Norflex)
- Drugs which slow the breakdown of dopamine (by inhibiting the enzyme catecholamine-o-methyl transferase):
- Entacapone (Comtan)
- Tolcapone (Tasmar)
- Others:
- The antidepressant amitriptyline (Elavil) is sometimes useful for fragmented sleep.
- Baclofen (Lioresal) is a muscle relaxant that may be useful in treating dystonic cramping during sleep.
Surgical Treatments
Various types of brain surgery have been tried, and continue to be used for intractable parkinsonism that cannot be controlled with medication. Although this is a radical step, it leaves the intellect intact and is better than complete incapacitation.
- Thalomotomy – Stereotaxic surgery (surgery through a small hole in the skull, using CT guidance for instrumentation) that destroys a part of the thalamus, performed in order to control tremors.
- Pallidotomy – Stereotaxic surgery that destroys a part of the basal ganglia known as the globus pallidus; performed in order to control abnormal movement (dyskinesia).
- Brain tissue transplantation – Insertion of human or animal fetal brain tissue into the area of the patient’s damaged basal ganglia, in the hope the new cells will produce dopamine.
- Deep brain stimulation – Permanent brain electrodes are placed, and electrical impulses delivered to control tremor (thalamus) or dyskinesia and stiffness (globus pallidus).
- Radiosurgery – Uses a linear accelerator or “gamma knife” to put precise beams of radiation on deep brain structures that cannot be reached by conventional brain surgery. Although this technique has been a useful technique for treating brain tumors, it has not produced encouraging results in the treatment of parkinsonism.
Continue to How to Get Disability Benefits for Parkinson’s Disease by Meeting a Listing.
